ABOUT US
GET A FREE QUOTE
CONTACT INFO
- +86-19941692089
- sales@suntramedical.com
- +86 19941692089
- Location: Room 201-235, Building 21, No. 369, Lushan Road, High-Tech Zone, Suzhou, China.
-
Home
-
Products
-
Hospital Furniture
-
Overbed Table
- OB-68 PP Overbed Table With Wheels
-
Parameters
Features
Size: L900*W450*H750-1100mm
Height adjustable by hydraulic
Equipped with 4 castors, 2 with brake
Tabletop made of PP material
Support frame made of aluminum, base is powder coated steel
Related Products
SMT-050DE Double Column Digital Radiography System (Electric)
Double column Digital Radiography System adopts large span independent ball tube frame design, easy installation, small occupation area, convenient maintenance. Four-way floating bed surface, easy to use and positioning. The lifting and lifting of X-ray source and bidirectional follow-up of horizontal position of bed column can reduce the positioning time and meet the requirements of large-flow clinical examination.
Applicable to orthopedics, emergency room, operating room, etc.,As human head, limbs, chest, spine, lumbar spine for medical diagnosis
AED-5 Automated External Defibrillator
1. 2 languages to choose from (by end user)
2. Bluetooth for data transmission (standard)
3. Daily, weekly and monthly self test
4. Use of AED in pocket (does not need to be opened)
5. Extra pocket in the bag for first aid products
6. IPX55 water and dust resistant
7. 5 volume levels (by the end user)
8. Button for adult/child mode
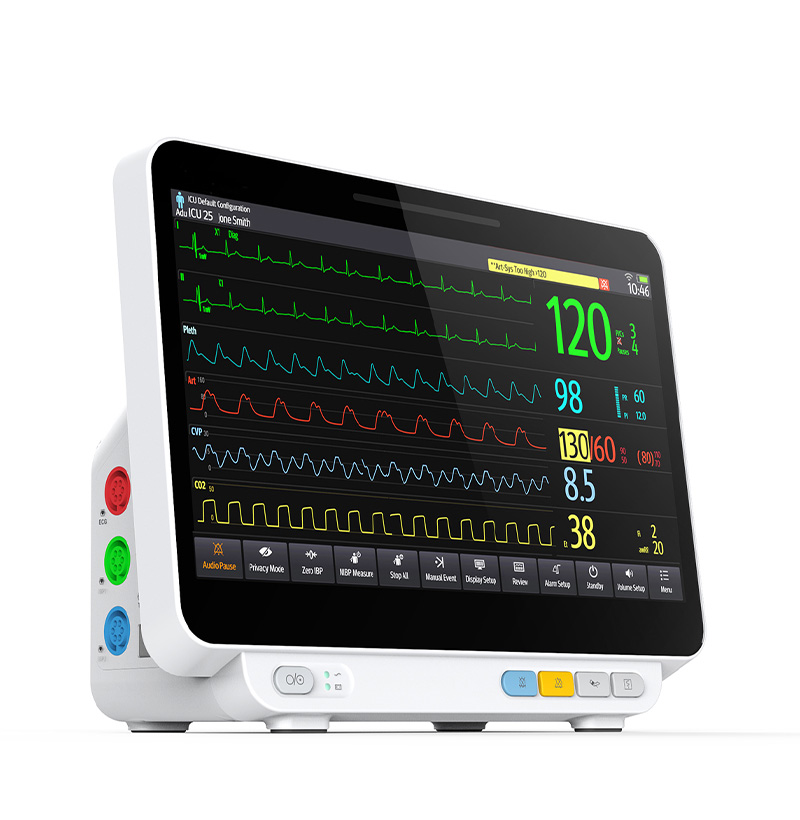
SM-8000D Multiparameter Patient Monitor
14”TFT LCD screen
SMT-40E Medical Ventilator
No external gas source can be independently long-term work (Bring your own air)
The operator control panel is with clear LCD display. Easy to operate.
Whole body is compact, easy to install on a ventilator tower crane, ambulance.
Friendly interface, easy operation and intuitive.
Offers three power: AC, Car Power, Built-in battery
Self-powered support ventilator work more than two hours.
SMH-60B 5-part Auto Hematology Analyzer
1. Throughput 60T/H.
2. 14-inch touch screen.
3. Tri-angle laser scatter + flow cytometry method + impedance method for RBC and PLT counting.
4. 3D holographic scattergram displays the accurate 5-part differentiation of WBC.
5. Large storage capacity: 300,000 results(including histogram, scattergram, patient information).
OUR LATEST
Our Latest Blog & News

-
7,April
Suntra Medical at CMEF 2026 Shanghai, China
Suntra Medical at CMEF 2026 Shanghai – Connecting Global Healthcare Partners
Shanghai, China – April 9-12, 2026
Suntra Medical is pleased to announce our active participation in the 93rd China International Medical Equipment Fair (CMEF 2026) , one of the world’s largest and most influential medical equipment exhibitions.
As a dedicated one-stop medical equipment sourcing provider, we are leveraging this premier industry event to connect with leading manufacturers, stay at the forefront of emerging technologies, and strengthen our global supply chain network. Our team is on the ground, engaging with industry partners and identifying cutting-edge solutions across key product categories including:
– Ultrasound systems
– Patient monitors
– Infusion pumps
– Anesthesia machines
– X-ray equipment
– ICU equipment
Contact us during CMEF 2026 (April 9-12) via:
WhatsApp / WeChat: +86 19941692089
Email: sales@suntramedical.com
Website: www.suntramedical.com

-
8,December
Suntra Launches Advanced Operating Theater Integration Solutions
*July 25, 2024 – Suzhou, China*
Suntra Medical unveils its comprehensive operating room integration system, designed to streamline surgical workflow and enhance operation theater efficiency. The integrated solution combines surgical lighting systems, operating tables, and advanced visualization equipment into a unified platform. Featuring 4K imaging technology and ergonomic design, these operating theater solutions reduce equipment setup time by 30% and improve surgical team coordination. The system’s modular architecture allows for customized configurations to meet specific surgical specialty requirements. This innovation reinforces Suntra’s position as a premier medical device manufacturer providing cutting-edge operating room equipment for modern healthcare facilities seeking to optimize their surgical environment and patient outcomes.

-
8,December
Digital X-Ray vs Conventional X-Ray: Key Differences for Medical Facilities
*July 22, 2024 – Suzhou, China
Understanding the transition from conventional radiography to digital x-ray systems is crucial for modern healthcare facilities. Suntra Medical, a trusted medical imaging equipment supplier, highlights the critical differences:
Image Quality & Processing: While traditional x-ray machines rely on film development, our digital radiography systems provide immediate high-resolution images with superior contrast resolution, enabling better detection of pulmonary diseases and bone fractures.
Radiation Safety: Digital x-ray equipment reduces patient exposure by up to 80% compared to conventional radiography, addressing growing concerns about radiation protection in medical imaging.
Workflow Efficiency: The digital imaging systems eliminate film processing time and integrate seamlessly with PACS and hospital information systems, significantly improving diagnostic workflow in busy medical facilities.
Cost Considerations: Although digital x-ray price is higher initially, the elimination of film and chemical costs provides better long-term ROI for hospital budgeting.

-
8,December
Emerging Markets Fuel Demand for Affordable and Localized Medical Technology Solutions
June 18, 2024 – Global Medical Device Markets
Growing healthcare needs across Southeast Asia, Africa, and Latin America are driving medical device manufacturers to develop cost-effective medical equipment tailored to resource-limited settings. These products retain core functionalities while incorporating simplified designs, localized manufacturing, and durable components to reduce costs and enhance accessibility. This trend not only addresses gaps in healthcare equity but also opens new growth avenues for companies specializing in emerging market medical solutions. Key segments seeing rapid adoption include portable diagnostic devices, reusable surgical instruments, and mobile clinic setups, all designed to perform reliably in diverse environments and supply chain conditions.
-
8,December
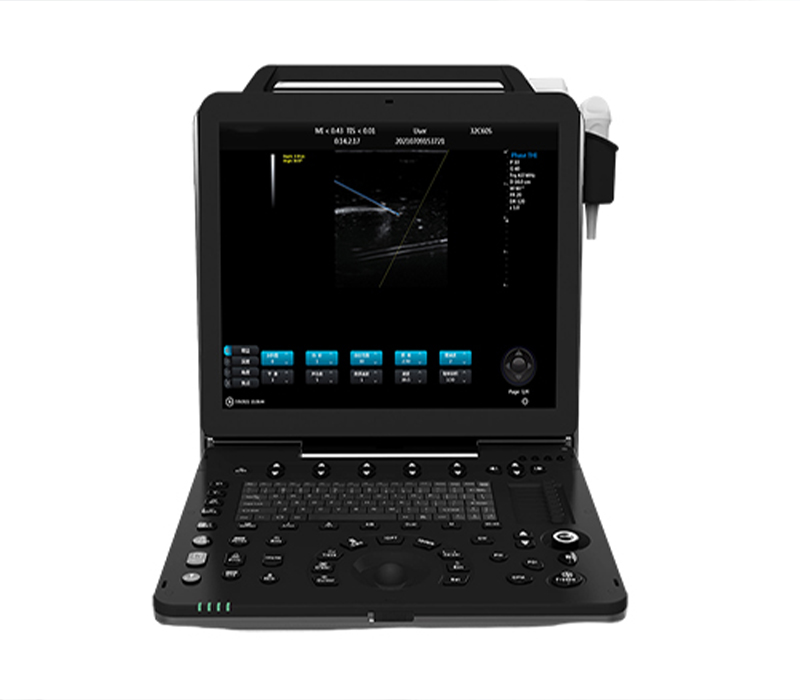
Portable Ultrasound Systems Gain Traction as First-Line Tool for Point-of-Care Diagnostics
*July 8, 2024 – Medical Imaging & Point-of-Care Ultrasound*
Handheld ultrasound devices are transforming clinical workflows by serving as the primary imaging tool in emergency medicine, primary care clinics, and remote medical settings. Advances in portable ultrasound technology, combined with AI-assisted image interpretation, have made these devices more intuitive and accessible for non-specialist clinicians. These point-of-care ultrasound (POCUS) systems support faster decision-making in critical scenarios such as trauma, respiratory distress, and abdominal emergencies. With the global shift toward decentralized healthcare, compact ultrasound machines are poised to become essential equipment in improving diagnostic speed and accuracy outside traditional radiology departments.

-
8,December
Telemedicine Adoption Grows 150% for Chronic Disease Management, Study Reveals
May 20, 2024 – Telemedicine & Digital Health
According to a recent study published in the Journal of Medical Internet Research, the use of telemedicine for chronic disease management has surged by 150% in the post-pandemic period. The analysis, which included over 100,000 patients, confirmed that remote patient monitoring and virtual consultations significantly improve medication adherence, patient engagement, and overall satisfaction. Despite this rapid growth, key barriers remain, including limited digital literacy among elderly patients and inconsistent telemedicine reimbursement policies. The findings underscore the importance of integrating scalable telehealth platforms and remote care solutions into mainstream healthcare delivery, particularly for managing conditions such as diabetes, hypertension, and COPD.
SERVICES
Services

Warranty Term and Technical Support
1. Two-Year Comprehensive Warranty
Suntra Medical provides a standard two-year warranty for all main product units, commencing from the production date. This warranty covers free repair services for any malfunction resulting from manufacturing or quality defects during the coverage period.
2. Value-Added Upgrade Services
To help our customers stay current with evolving needs, Suntra may offer functional upgrades during the product lifecycle. These upgrades—which may enhance performance, expand application scope, or improve functionality—are model-specific and subject to product compatibility.
3. Streamlined Warranty Process
For validated warranty claims requiring part replacement, Suntra will ship new replacement components at our expense. Following installation, customers are responsible for returning the defective parts to Suntra using provided shipping instructions.
4. Post-Warranty Support
For services beyond the standard warranty period, we provide detailed cost estimates and proforma invoices based on actual repair costs. All repairs are conducted after payment confirmation and include rigorous testing to ensure optimal performance before returning to the customer.
5. Long-Term Technical Commitment
Suntra ensures your operational continuity through comprehensive technical support including remote troubleshooting, detailed documentation, and operator training. We guarantee spare parts availability for a minimum of 8 years after delivery, protecting your investment and minimizing operational downtime.

OEM/ODM Solutions and Services
Suntra recognizes that standard products may not always address specific market needs. Our integrated OEM (Original Equipment Manufacturing) and ODM (Original Design Manufacturing) services enable complete product customization—from conceptual design and engineering development to final manufacturing and branding. Whether adapting an existing model or creating an entirely new device, we collaborate closely to deliver solutions that reflect your unique requirements and vision.
1. Brand Integration Solutions
We offer complete white-label solutions, allowing you to market products under your own brand while we handle manufacturing and quality assurance.
2. Custom Branding & Marking
Customize product surfaces, panels, and packaging with your logos, colors, and identifiers to maintain brand consistency across all touchpoints.
3. Component Supply Services
We supply individual components and replacement parts for integration into your own products or for after-sales service requirements.
4. Tailored Manufacturing Services
From modified existing models to fully original designs, we realize your specifications through flexible engineering and scalable production.

Regulatory Consulting & Certification Support
Navigating international standards and certifications presents a significant challenge in entering new markets. Suntra simplifies this complexity through specialized regulatory consulting and end-to-end certification support.
We provide targeted guidance on key compliance requirements for your target regions and assist in preparing technical documentation, coordinating with accredited bodies, and managing the complete approval process. This integrated service saves valuable time, reduces compliance risks, and ensures your projects meet all necessary regulatory standards.

Customer Collaboration Process
At Suntra, we build successful partnerships through transparent communication and well-defined expectations. Our streamlined collaboration process ensures efficiency and clarity at every stage.
1. Inquiry & Consultation
Share your requirements with our team, and we will assign a dedicated project manager to gain a comprehensive understanding of your needs.
2. Proposal & Quotation
Receive a detailed technical proposal with transparent pricing and commercial terms tailored to your project.
3. Order Confirmation & Payment
After quotation approval, we issue a proforma invoice. Order confirmation is completed upon receipt of your signed PI, with production commencing following advance payment.
4. Production & Quality Assurance
Your order enters our manufacturing schedule, undergoing rigorous quality control checks throughout each production phase.
5. Delivery & After-Sales Support
We coordinate secure shipment with complete documentation, followed by comprehensive after-sales service and ongoing technical support.